
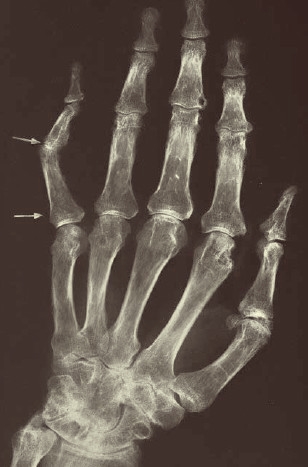
Fig. 9.1 Rheumatoid arthritis with joint swelling and moderate ulnar deviation of the fingers.
Rheumatoid Arthritis
BASIC INFORMATION
DEFINITION
Rheumatoid arthritis (RA) is a systemic disorder characterized by chronic joint inflammation that most commonly affects peripheral joints. This process results in the development of pannus, a destructive tissue that damages cartilage. Rheumatoid arthritis is the most frequent inflammatory rheumatic joint disease. Women are affected about three times more often than men.
Clinical Findings
Symmetric distribution of the joint disorder is characteristic. In the early phase of the disease hand, metacarpophalangeal and interphalangeal joints (Fig. 9.1), as well as knee and metatarsophalangeal joints, are affected. Involvement of the distal interphalangeal joints is rare and points to differential diagnoses, such as psoriatic arthritis or reactive arthritis. Usual symptoms of rheumatoid arthritis include joint pain and swelling, often associated with severe and long-standing morning stiffness, as well as loss of strength, particularly in the hands. Fatigue and general malaise, and at times slightly elevated temperatures, are often the first indications of the ongoing disease process. Without effective medication, rheumatoid arthritis is characterized by functional deficit due to progressive joint destruction. Late stages are characterized by deformities, rheumatic nodules, as well as postinflammatory changes of bones, joints, and soft tissue. In later stages of the disease extra-articular manifestations may occur. This includes pleuropericarditis, rheumatic nodules, eye involvement, and more rarely, vasculitis with sensorimotor disturbances or amyloidosis.
EPIDEMIOLOGY & DEMOGRAPHICS
PREVALENCE: 5 cases/1000 adults
PREVALENT AGE: 35 to 45 yr
PREDOMINANT SEX:
• Female: male ratio of 3:1
• After age 50 yr, sex difference less marked
PHYSICAL FINDINGS & CLINICAL PRESENTATION
• Usually gradual onset; common prodromal symptoms of weakness, fatigue, and anorexia
• Initial presentation: multiple symmetric joint involvement, most often in the hands and feet, usually MCP, MTP, and PIP joints
Fig. 9.2 Rheumatoid arthritis: considerable erosions, joint space narrowing, and periarticular osteopenia.
• Joint effusions, tenderness, and restricted motion usually present early in the disease
• Eventual characteristic deformities: subluxations, dislocations, and joint contractures
• Extraarticular findings:
1. Tendon sheaths and bursae frequently affected by chronic inflammation
2. Possible tendon rupture
3. Rheumatoid nodules over bony prominences such as the elbow and shaft of the ulna
4. Splenomegaly, pericarditis, and vasculitis
5. Findings of carpal tunnel syndrome resulting from flexor tenosynovitis
• Eventual characteristic deformities: subluxations, dislocations, and joint contractures
• Extraarticular findings:
1. Tendon sheaths and bursae frequently affected by chronic inflammation
2. Possible tendon rupture
3. Rheumatoid nodules over bony prominences such as the elbow and shaft of the ulna
4. Splenomegaly, pericarditis, and vasculitis
5. Findings of carpal tunnel syndrome resulting from flexor tenosynovitis
ETIOLOGY
Unknown. There is increasing evidence that the inflammation and destruction of bone and cartilage that occurs in many rheumatic diseases are the result of the activation by some unknown mechanism of proinflammatory cells that infiltrate the synovium. These cells, in turn, release various substances, such as cytokines and tumor necrosis factor (TNF) alpha, which subsequently cause the pathologic changes typical of this group of diseases. Many of the newer therapeutic agents are directed at the suppression of these final mediators of inflammation.
Unknown. There is increasing evidence that the inflammation and destruction of bone and cartilage that occurs in many rheumatic diseases are the result of the activation by some unknown mechanism of proinflammatory cells that infiltrate the synovium. These cells, in turn, release various substances, such as cytokines and tumor necrosis factor (TNF) alpha, which subsequently cause the pathologic changes typical of this group of diseases. Many of the newer therapeutic agents are directed at the suppression of these final mediators of inflammation.
DIAGNOSIS
Already in the early course of the disease radiologic changes may be detected in the hands and feet. In the early stage changes include periarticular soft tissue swelling and demineralization of the periarticular bones. In later stages they include joint space narrowing along with erosions and subluxations (Fig. 9.2). It is rare for ankylosis to be a feature. Involvement of the cervical spine however is frequent. Rheumatoid arthritis may lead to spondylarthritis, instability, or rarely ankylosis, but also to destruction of the atlantal dentate ligaments through inflammatory pannus resulting in atlantoaxial subluxation or even compression of the spinal cord. Laboratory examination often reveals elevated erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), normochromic and normocytic anemia, thrombocytosis, and low serum iron. Rheumatoid factors tend to be positive at later stages.
DIFFERENTIAL DIAGNOSIS
• SLE
• Seronegative spondyloarthropathies
• Polymyalgia rheumatica
• Acute rheumatic fever
• Scleroderma
According to the American College of Rheumatology, RA exists when four of seven criteria are present, with criteria 1 to 4 being present for at least 6 wk.
1. Morning stiffness over 1 hr
2. Arthritis in three or more joints with swelling
3. Arthritis of hand joints with swelling
4. Symmetric arthritis
5. Rheumatoid nodules
6. Roentgenographic changes typical of RA
7. Positive serum rheumatoid factor
LABORATORY TESTS
• Increase in rheumatoid factor in 80% of cases (rheumatoid factor also present in the normal population)
• Possible mild anemia
• Usually, elevated acute phase reactants (ESR, C-reactive protein)
• Possible mild leukocytosis
• Usually, turbid joint fluid, which forms a poor mucin clot; elevated cell count, with an increase in polymorphonuclear leukocytes
IMAGING STUDIES
Plain radiography
• Usually reveals soft-tissue swelling and osteoporosis early
• Eventually, joint space narrowing, erosion, and deformity visible as a result of continued inflammation and cartilage destruction
TREATMENT
NONPHARMACOLOGIC THERAPY
Proper management requires close cooperation among primary physician, therapist, rheumatologist, and orthopedist.
• Patient education is important.
• Rest with proper exercise and splinting can prevent or correct joint deformities.
• Maintain proper diet and control obesity.
CHRONIC Rx
• NSAIDs: commonly used as the initial treatment to relieve inflammation (drug of choice for most patients: aspirin, but other NSAIDs also effective)
• Disease-modifying drugs (DMARDs): are traditionally begun when NSAIDs are not effective; current recommendations favor early aggressive treatment with
DMARDs, seeking to minimize long-term joint damage. Commonly used agents are methotrexate, cyclosporine, hydroxychloroquine, sulfasalazine, leflunomide, and infliximab. Most of these are associated with potential toxicity and require close monitoring. They are also usually slow-acting drugs that require more than 8 wk to become effective (see Table 1-13)
• Oral prednisone
• Intrasynovial steroid injections
• Etanercept (Enbrel), a tumor necrosis factor alpha blocker is indicated in moderately to severely active RA in patients who respond inadequately to DMARDs. The combination of etanercept and methotrexate has been reported to be effective and promising in the treatment of RA.
DISPOSITION
• Remissions and exacerbations are common, but condition is chronically progressive in the majority of cases.
• Joint degeneration and deformity often lead to disability.
• Early diagnosis and treatment are important and can improve quality of life.
Already in the early course of the disease radiologic changes may be detected in the hands and feet. In the early stage changes include periarticular soft tissue swelling and demineralization of the periarticular bones. In later stages they include joint space narrowing along with erosions and subluxations (Fig. 9.2). It is rare for ankylosis to be a feature. Involvement of the cervical spine however is frequent. Rheumatoid arthritis may lead to spondylarthritis, instability, or rarely ankylosis, but also to destruction of the atlantal dentate ligaments through inflammatory pannus resulting in atlantoaxial subluxation or even compression of the spinal cord. Laboratory examination often reveals elevated erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), normochromic and normocytic anemia, thrombocytosis, and low serum iron. Rheumatoid factors tend to be positive at later stages.
DIFFERENTIAL DIAGNOSIS
• SLE
• Seronegative spondyloarthropathies
• Polymyalgia rheumatica
• Acute rheumatic fever
• Scleroderma
According to the American College of Rheumatology, RA exists when four of seven criteria are present, with criteria 1 to 4 being present for at least 6 wk.
1. Morning stiffness over 1 hr
2. Arthritis in three or more joints with swelling
3. Arthritis of hand joints with swelling
4. Symmetric arthritis
5. Rheumatoid nodules
6. Roentgenographic changes typical of RA
7. Positive serum rheumatoid factor
LABORATORY TESTS
• Increase in rheumatoid factor in 80% of cases (rheumatoid factor also present in the normal population)
• Possible mild anemia
• Usually, elevated acute phase reactants (ESR, C-reactive protein)
• Possible mild leukocytosis
• Usually, turbid joint fluid, which forms a poor mucin clot; elevated cell count, with an increase in polymorphonuclear leukocytes
IMAGING STUDIES
Plain radiography
• Usually reveals soft-tissue swelling and osteoporosis early
• Eventually, joint space narrowing, erosion, and deformity visible as a result of continued inflammation and cartilage destruction
TREATMENT
NONPHARMACOLOGIC THERAPY
Proper management requires close cooperation among primary physician, therapist, rheumatologist, and orthopedist.
• Patient education is important.
• Rest with proper exercise and splinting can prevent or correct joint deformities.
• Maintain proper diet and control obesity.
CHRONIC Rx
• NSAIDs: commonly used as the initial treatment to relieve inflammation (drug of choice for most patients: aspirin, but other NSAIDs also effective)
• Disease-modifying drugs (DMARDs): are traditionally begun when NSAIDs are not effective; current recommendations favor early aggressive treatment with
DMARDs, seeking to minimize long-term joint damage. Commonly used agents are methotrexate, cyclosporine, hydroxychloroquine, sulfasalazine, leflunomide, and infliximab. Most of these are associated with potential toxicity and require close monitoring. They are also usually slow-acting drugs that require more than 8 wk to become effective (see Table 1-13)
• Oral prednisone
• Intrasynovial steroid injections
• Etanercept (Enbrel), a tumor necrosis factor alpha blocker is indicated in moderately to severely active RA in patients who respond inadequately to DMARDs. The combination of etanercept and methotrexate has been reported to be effective and promising in the treatment of RA.
DISPOSITION
• Remissions and exacerbations are common, but condition is chronically progressive in the majority of cases.
• Joint degeneration and deformity often lead to disability.
• Early diagnosis and treatment are important and can improve quality of life.
| TYPE/GENERIC (TRADE) NAME | RECOMMENDED DOSAGES | TOXIC EFFECTS | RECOMMENDED MONITORING |
| Gold compounds (Myochrysine) | IM: 10 mg followed by 25 mg 1 wk later, then 25-50 mg wkly until there is toxicity, major clinical improvement, or cumulative dose = 1 g. If effective, interval between doses is increased. | Pruritus, dermatitis, stomatitis, nephrotoxicity, blood dyscrasias, “nitritoid” reaction: flushing, weakness, nausea, dizziness 30 min after injection. | CBC, platelet count before every other injection. |
| Aurothioglucose (Solganal) | IM: 10 mg; 2nd and 3rd doses 25 mg, 4th and subsequent 50 mg. Interval between doses: 1 wk. If improvement, no toxicity, decrease dose to 25 mg or increase interval between doses. | Dermatitis, stomatitis, nephrotoxicity, blood dyscrasias. | CBC, platelet count every 2 wk. |
| Auranofin (Ridaura) | Oral: 3 mg bid or 6 mg qd. May increase to 3 mg tid after 6 months. | Loose stools, diarrhea (up to 50%), dermatitis. | Baseline CBC, platelet count, U/A, renal, liver function, at onset then CBC with platelet count, U/A 9 months. |
| Antimalarial Hydroxychloroquine (Plaquenil) | Oral: 400-600 mg qd with meals then 200-400 mg qd. | Retinopathy, dermatitis, muscle weakness, hypoactive DTRs, CNS. | Ophthalmologic examination every 3 months (visual acuity, slitlamp, funduscopic, visual field tests), neuromuscular examination. |
Penicillamine (Cuprimine, Depen) |
Oral: 125-250 mg qd, then increasing at monthly intervals doses to max 750-1000 mg by 125-250 mg. |
Pruritus, rash/mouth ulcers, bone marrow depression, proteinuria, hematuria, hypogeusia, myesthenia, myositis, GI distress, pulmonary toxicity, teratogenic. | CBC every 2 weeks until dose stable, then every month. U/A weekly until dose stable, then every month. HCG as needed. |
| Methotrexate (Rheumatrex) | Oral: 7.5-15 mg weekly. | Pulmonary toxicity, ulcerative stomatitis, leukopenia, thrombocytopenia, GI distress, malaise, fatigue, chills, fever, CNS, elevated LFTs/liver disease, lymphoma, infection. | CBC with platelet count, LFTs weekly x 6 wk then monthly LFTs, U/A periodically, HCG as needed. |
| Azathioprine (Imuran) | Oral: 50-100 mg qd, increase at 4-wk intervals by 0.5 mg/kg/d up to 2.5 mg/kg/d. |
Leukopenia, thrombocytopenia, GI, neoplastic if previous Rx with alkylating agents. |
CBC with platelet count, wkly x 1 mo, 2x/mo. x 2 mo, then monthly, HCG as needed. |
| Sulfasalazine (Azulfidine) | Oral: 500 mg daily then increase up to 3 g daily. | GI, skin rash, pruritus, blood dyscrasias, oligospermia. |
CBC, U/A q 2 wk x 3 mo, then monthly x 9 mo, then every 6 mo. |
| Alkylating agents Cyclophosphamide (Cytoxan) | Oral: 50-100 mg daily up to 2.5 mg/kg/d. | Leukopenia, thrombocytopenia, hematuria, GI, alopecia, rash, bladder cancer, non-Hodgkin’s lymphoma, infection. | CBC with platelet count, regularly. HCG as needed. |
| Chlorambucil (Leukeran) | Oral: 0.1-0.2 mg/kg/d. | Bone marrow suppression, GI, CNS, infection. |
CBC with platelet count every wk. WBCs 3-4 days after each CBC during 1st 3-6 wk at therapy. HCG as needed. |
| Cyclosporine (Sandimmune) | Oral 2.5-5 mg/kg/d. | Nephrotoxicity, tremor, hirsutism, hypertension, gum hyperplasia. |
Renal function, liver function. |
| Pyrimidine, synthesis inhibitors | Loading dose: 100 mg/d for 3 days. | Hepatotoxicity, carcinogenesis. | LFTs every month, drug levels after discontinuation (after 1 month therapy, remains in blood for 2 years without use of cholestyramine). |
| Leflunomide (Arava) | Maintenance therapy: 20 mg/d; if not tolerated, 10 mg/d. |
Immunosuppression, long half-life. |
TABLE 1-13 -- Selected Disease-Modifying Antirheumatic Drugs
Contacts: lubopitno_bg@abv.bg www.encyclopedia.lubopitko-bg.com Corporation. All rights reserved.
DON'T FORGET - KNOWLEDGE IS EVERYTHING!